Levitra Or Vivanza? This Is How You Buy Vardenafil According EU Import Regulations
Variety of Vardenafil Brand Names
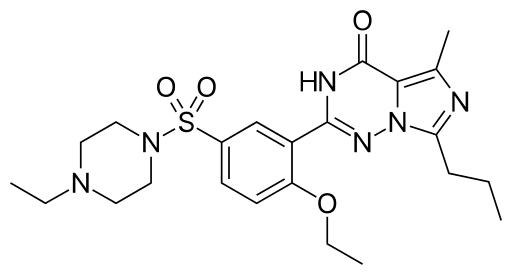
Vardenafil is an active substance falling in the group of strong PDE5 inhibitors treating erectile dysfunction.
 It has the following brands:
It has the following brands:
- Levitra produced by Bayer, Schering-Plough, GlaxoSmithKline;
- Vivanza brand is used for vardenafil distribution in Italy;
- Staxyn produced by pharmaceutical manufacturers in India.
What is the reason for appearing of so many trade names in global market? First vardenafil was marketed by a cooperation of pharmaceutical companies from Germany, the UK and the USA (Bayer, GlaxoSmithKline, and Schering-Plough accordingly) under Levitra brand name. Later on, the rights were returned to Bayer to promote vardenafil under Levitra name in divers countries outside the United States. Nevertheless, GlaxoSmithKline chose for vardenafil another brand name to sell in in Italy – Vivanza. Hence, due to trade rules of EU, Levitra is sold in the immediate neighborhood to Vivanza resulting from parallel import. Canada and the United States have approved the tableted forms branded as Levitra Soft and Staxyn.
All Levitra (Vardenafil) information in this article: mycanadianpharmacyrx.com/generic-levitra
Vardenafil Characteristics and Pharmacology
This is a phosphodiesterase type 5 inhibitor. Vardenafil hydrochloride is almost a colorless solid substance. Its pharmacological effect is aimed at improvement of erectile function. Erection is a hemodynamic process based on relaxation of unstriated muscles of cavernous bodies and arterioles associated with them. Sexual stimulation releases nitrogen oxide (which activates guanylate cyclase causing increased synthesis of cyclic guanosine monophosphate in smooth cells of cavernous bodies) from endothelial cells of cavernous bodies. Accumulation of cyclic guanosine monophosphate leads to relaxing of smooth muscle cells and cavernous bodies trabecula, to increase of blood stream to penis and finally – erection.
Since sexual stimulation is important for initiation of the process of local release of nitrogen oxide, PDE5 inhibition doesn’t have any effect without sexual stimulation. Vardenafil activity in regards to PDE5 surpasses the one of other well-known phosphodiesterase isoforms.
Vivanza/Levitra Pharmacokinetics
Vardenafil pharmacokinetics is almost proportional to the dose in recommended range. It is fast absorbed inside. The absolute bioaccessibility is 15 per cent. In case of intake by sound volunteers of one-time dosage of 20mg, time to reach maximum concentration in plasma is 30-120 minutes (in average – 60 minutes). In case of simultaneous intake with fat food, maximal concentration decreases by 18-50 per cent (as per results of 2 research studies). Volume of vardenafil distribution at steady state is 208l which indicates to extensive distribution in all tissues.
Vardenafil and its main circulating metabolite (M1) is well connected with serum proteins (around 95%). The connection is reversible and doesn’t depend on overall concentration of medicinal product. Healthy volunteers observed the one-time dosage of vardenafil (20mg) emitted in semen in 1,5 hours after the intake.
Vivanza/Levitra Advantages
Vardenafil is as ten times as more efficient as sildenafil and as thirteen times as tadalafil.
Its effectiveness was proved by German scientists.

- Vardenafil takes its action much earlier than the analogues. There are cases, when the effect occurred within 10 minutes after the intake. Other medications require at least 25 minutes.
- Effect duration of Levitra/Vivanza is not so long as Cialis’s one, nonetheless 4-6 hours is sufficient period of time for sexual intercourse.
- Vardenafil causes significantly less side-effects (they occur only in 2% of cases).
- German and English scientists have demonstrated that Levitra/Vivanza is efficient in 50% of cases when Viagra is not valid.
- Vardenafil proves to be effective among 70% of men who underwent prostatic gland removal and among 72% of men suffering from diabetes.
- Moreover, according to the recent research studies in the U.S., it was shown that after taking 10mg of Levitra/Vivanza, patients could increase the duration of sexual intercourse from 6 to 12 minutes without any adverse effects.
All these medical trials confirm high effectiveness of the medication.
Vardenafil Counter-Indications
Vardenafil products have the following contraindications:
- Hypersensitivity to Vivanza or Levitra ingredients;
- Co-administration with nitrates or other medicinal products acting as nitrogen oxide donors (PDE5 inhibitors have hypotensive effect of nitrates; corresponding time interval between vardenafil and subsequent usage of nitrates or nitrogen oxide donors is not determined);
- Combination with alpha-adrenergic blocking agents (possible development of hypotension).
Vardenafil is not indicative to women, children and newly-born.
Vivanza/Levitra Possible Side-Effects
From the part of body as a whole: anaphylactic reactions (including swelling of the larynx), asthenia, face edema, pain.
From the part of acoustic apparatus: tinnitus aurium.
From the part of cardio-vascular system: cardiac angina, chest pains, arterial hypertension/hypotension, myocardial ischemia, myocardial infarction, heartbeats, postural hypotension, syncopal conditions, tachycardia.
From the part of gastro-intestinal tract: abdominal pain, changing of functional hepatic tests, dryness in mouth, dysphagy, esophagitis, gastritis, esophageal reflux, increase of the level of gamma- glutamyl transpeptidase, vomit.
From the part of supporting-motor apparatus: arthralgia, pain in neck, myodynia, backache.
From the part of nervous system: hypesthesia, paresthesia, insomnia, drowsiness, vertigo.
From the part of respiratory system: dyspnea, nasal bleeding, pharyngitis.
From the part of skin covering: photosensitivity, rashes, itching, sweating.
From the part of visual organs: impairment of vision, blurring of vision, chromatic sensitivity changes, conjunctivitis, weak vision, glaucoma, photophobia.
From the part of urinary tract: ejaculation impairment, priapism (including prolonged and painful erection).
Interactions
Cytochrome P450 inhibitors
- Cimetidine (400mg twice a day) didn’t take significant influence on bioaccessibility and maximal concentration of vardenafil in case of simultaneous intake by sound volunteers.
- Erythromycin (500mg 3 times per day) caused a four-time increase of bioaccessibility and a three-time increase of vardenafil concentration in case of combined 5mg intake by healthy volunteers.
- Ketoconazole (200mg once a day) caused a 10 times increase of vardenafil (5mg) bioaccessibility and a four-time increase of its maximal concentration. The dosage of 5mg vardenafil cannot be exceeded in case of combined daily intake with ketoconazole (200mg). Since larger dosages of ketoconazole (400mg daily) may lead to a significant increase of bioaccessibility and maximal concentration.
HIV protease inhibitors
- Indinavir (800mg three times a day) along with vardenafil (10mg) may cause a considerable increase of bioaccessibility, maximal concentration and elimination half-life of vardenafil. It is not recommended increasing one-time dose of vardenafil within the 24-hour period if co-administered with Indinavir.
- Ritonavir (600mg twice a day) along with vardenafil (5 mg) may lead to a measurable increase of bioaccessibility, maximal concentration and elimination half-life of vardenafil. Interaction is a consequence of hepatic metabolism blockage of vardenafil by Ritonavir.
Other Medications
There was no pharmacokinetic interaction between vardenafil and such medicinal products as glibenclamide, warfarin, digoxin, Maalox, and ranitidine.
Nitrates
Vardenafil (20mg) enhances hypotensive action of sublingual nitrates (0,4mg) in case of their intake in 1 and 4 hours after vardenafil usage and increases cardiac rhythm in 1,5 and 8 hours after vardenafil intake. Co-administration of vardenafil with nitrates is counter-indicative.
Procardia
The combined intake of vardenafil (20mg) along with Procardia of extended release, the latter hasn’t taken any impact on bioaccessibility, maximal concentration and elimination half-life of vardenafil. Procardia doesn’t change plasmatic level of vardenafil as well.
Alpha-adrenergic blocking agents
Patients treated with alpha-adrenergic blocking agents should avoid combining their administration with vardenafil. Their combination causes considerable hypotension.
Their combined intake with vardenafil (5mg) caused the increased indexes of Ritonavir and Indinavir bioaccessibility and maximal concentration.
Alcohol
Simultaneous intake of alcohol with vardenafil causes no changes in plasmatic levels of vardenafil and ethanol. Vardenafil hasn’t potentiated hypotensive action of alcohol among healthy volunteers within the four-hour observation.
Acetylsalicylic acid
Vardenafil (10 and 20mg) doesn’t cause the bleeding period caused by acetylsalicylic acid.
Other interactions
Vardenafil didn’t influence pharmacodynamics of glibenclamide (glucose concentration and insulin) and warfarin.
Vivanza/Levitra Overdose
Symptoms: reversible lower back pain, myodnia, blurred vision.
Treatment: supporting therapy. It is not expected that dialysis will prove effective due to increase of clearance, since vardenafil is characterized by high degree of connection with plasma proteins and is not cleared considerably with urine.